What the Latest NEJM Review Gets Right, and Glosses Over
A Critique and Summary of the GLP-1 Receptor Agonists Review by Clifford J. Rosen and Julie R. Ingelfinger, published in NEJM ON April 2, 2026.
The New England Journal of Medicine just published a sweeping review of GLP-1 receptor agonists, the class of drugs behind Ozempic, Wegovy, and Mounjaro, and it reads like a victory lap for one of modern medicine’s most dramatic pharmaceutical success stories. But beneath the impressive trial data lies a set of unanswered questions that clinicians and patients can’t afford to ignore.
What Are GLP-1 Receptor Agonists?
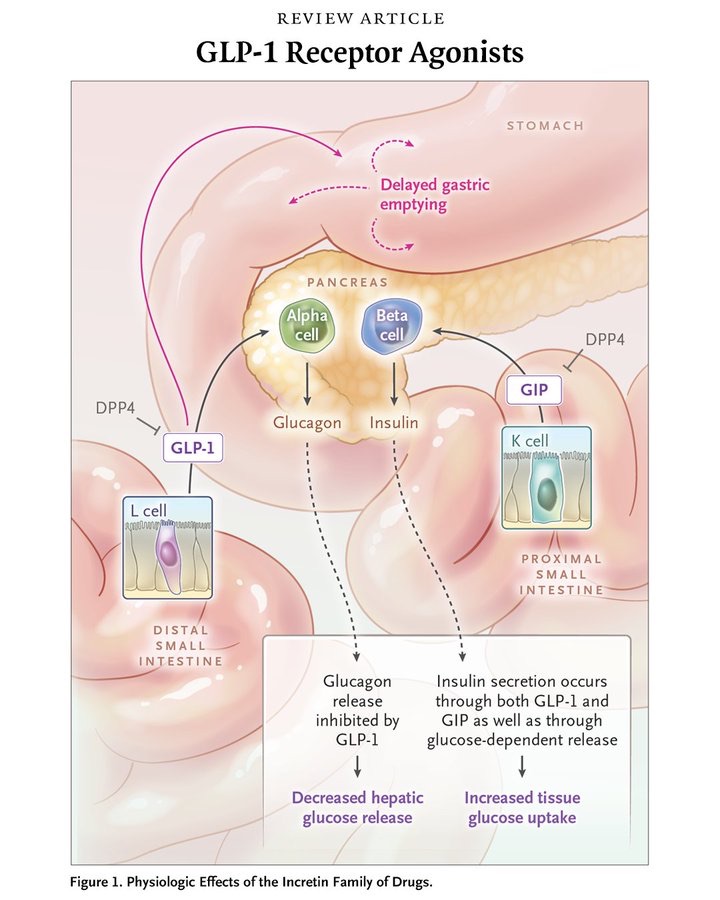
GLP-1 (glucagon-like peptide-1) receptor agonists are synthetic versions of gut hormones called incretins, which trigger insulin release after meals. They work through multiple pathways: slowing gastric emptying, suppressing glucagon, signaling satiety centers in the hypothalamus, and in newer dual/triple agonists also activating receptors for GIP (glucose-dependent insulinotropic peptide) and glucagon. The drug class traces its origins to a surprising source: Gila monster venom, from which researcher John Eng isolated a peptide called exendin-4 in 1990 that led directly to the first approved drug, exenatide, in 2005.
The Efficacy Data Is Impressive
The clinical trial evidence reviewed here is hard to argue with:
- Glucose control: Tirzepatide (Mounjaro), the dual GLP-1/GIP agonist, reduced HbA1c by up to 2 percentage points, with nearly 90% of participants hitting target glucose levels in the SURPASS-1 trial.
- Weight loss:** Semaglutide (Wegovy) produced an average 14.9% body weight reduction in the STEP 1 trial; tirzepatide beat that with 15–21% losses; and the next-gen triple agonist retatrutide hit nearly 25% weight loss in phase 2.
- Heart protection: In the SUSTAIN 6 trial, weekly semaglutide cut the risk of major cardiovascular events (heart attack, stroke, cardiovascular death) by 26% in high-risk diabetic patients.
- Kidney protection: A meta-analysis of major trials showed a composite kidney outcome — including renal failure was reduced by 18%, prompting FDA approval of semaglutide for kidney disease risk in January 2025.
- Liver disease: Both tirzepatide and retatrutide showed clinically significant resolution of metabolic dysfunction–associated liver disease (formerly called NAFLD).[1]
The Side Effect Picture Is Incomplete
The review dutifully catalogs known adverse events, but the framing is optimistic to a fault.
The most common issues are gastrointestinal — nausea, vomiting, diarrhea, constipation — occurring especially during dose escalation, with a 4–8% trial dropout rate.
More concerning findings include:
- Bone and muscle loss: A significant 1% loss of bone mineral density in the femur and spine was reported after just 52 weeks of liraglutide treatment, with similar signals from semaglutide. The article flags this but notes long-term fracture data simply don’t exist yet.
- Gallbladder disease: A meta-analysis of 76 trials and 100,000+ participants found 1.5x the risk of gallbladder and biliary disease with GLP-1 agonists vs. placebo.
- Pancreatitis: While large RCTs haven’t confirmed a clear signal, a real-world analysis of 16 million electronic health records showed higher pancreatitis cases with GLP-1 agonists.
- Wight regain: The STEP 4 trial showed clinically significant weight regain after stopping semaglutide. The implication, that patients may need these drugs indefinitely, is enormous for cost, access, and long-term safety.
The Health Equity Section Is the Conscience of the Paper
One of the review’s most important passages is also one of its most understated: the acknowledgment that Black and Hispanic patients are systematically underrepresented in GLP-1 trials, despite making up a disproportionate share of people with obesity and type 2 diabetes.
Only one substudy (from SURPASS-1) examined Hispanic responses specifically. The authors call for “price adjustments and plans for greater distribution,” but don’t grapple seriously with how a drug class priced at $900–$1,300/month gets equitably deployed.
What’s Coming: The Pipeline
The article includes a detailed table of drugs under development that signals where this field is heading:
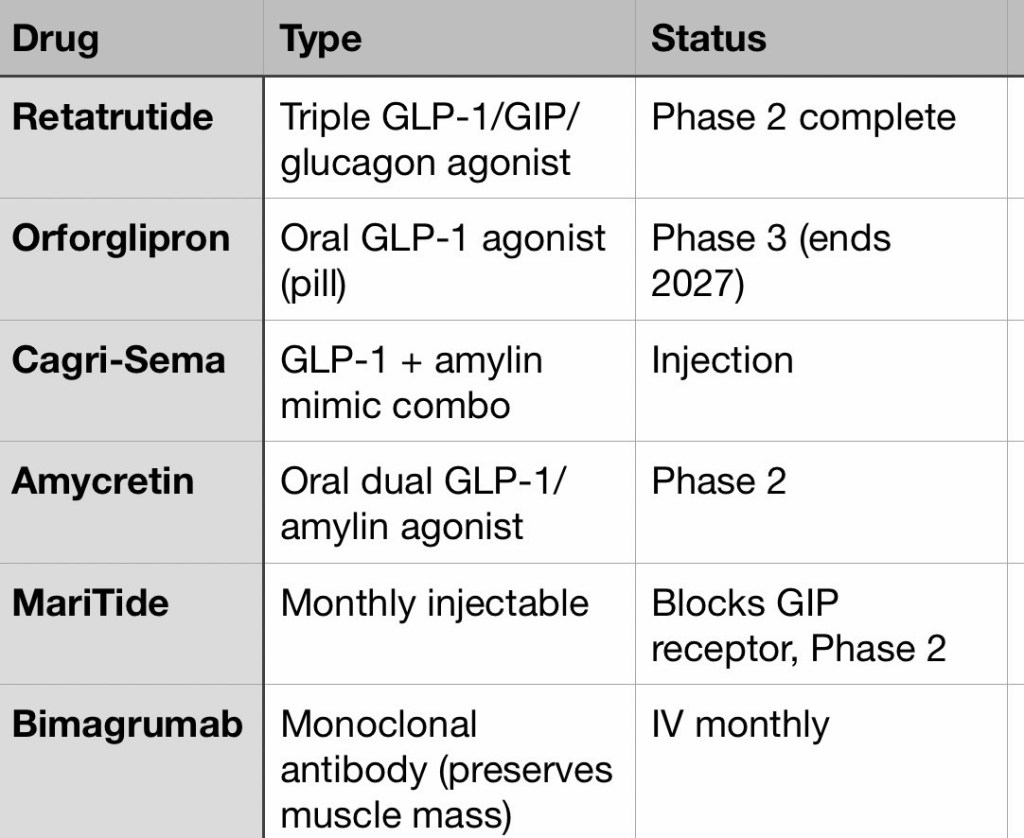
The move toward oral formulations (orforglipron, danuglipron, amycretin) and monthly injectables (MariTide) could dramatically change adherence and access.
Where the Review Falls Short
For all its comprehensiveness, this article has several notable blind spots worth flagging for a general audience:
- The “indefinitely or episodically?” question is raised but dropped. The review acknowledges no one knows whether these drugs should be taken for life, cycled, or microdosed and then moves on. For a drug that causes weight regain on cessation, this is the central clinical and ethical question.
- Cost is mentioned but not interrogated. The word “cost” appears a handful of times, mostly as a caveat. A paper in NEJM in 2026, when GLP-1 agonists have already created supply shortages, owes readers a more rigorous engagement with the economics.
- The muscle loss data gap is alarming. The review notes there are “few studies with data on long-term consequences” for muscle function and bone. Given that muscle loss is a known complication of all major weight-loss interventions and has serious implications for aging populations, this should be a red flag, not a footnote.
- Trial duration is short for a lifelong drug. The authors themselves note that the average duration of major cardiovascular outcome trials was only ~3 years. For drugs that may be taken for decades, this is a significant evidence gap.
Bottom Line
The NEJM review makes a compelling, evidence-based case that GLP-1 receptor agonists represent a genuine revolution in treating obesity and type 2 diabetes, with meaningful cardiovascular and renal benefits extending well beyond weight loss.
The next-generation triple agonists and oral formulations in the pipeline suggest the best may still be ahead. But the durability of benefit depends on long-term adherence, the safety profile over decades remains unknown, and the equity and cost problems threaten to make this revolution one that only the well-insured can afford to join.